AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Hydrogen bonds8/22/2023  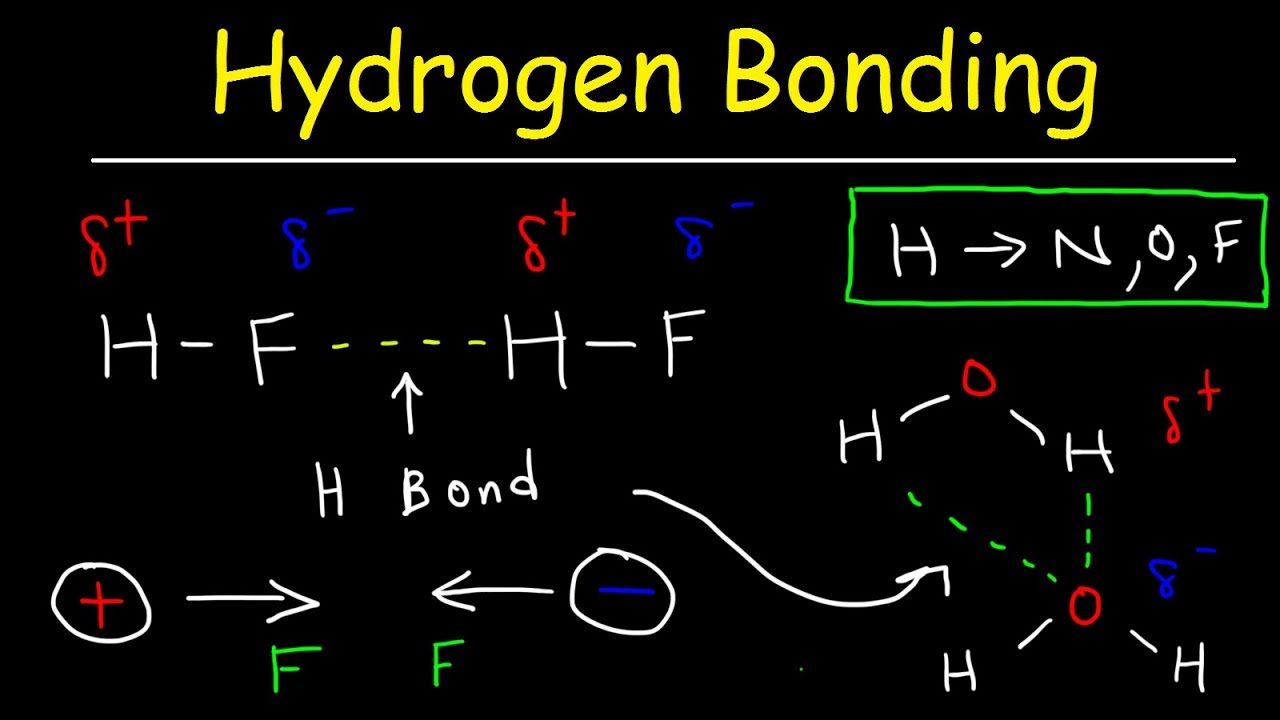
We can determine that ethanol can form a maximum of 3 hydrogen bond only, hence hydrogen bond is least extensive, least amount of energy required to overcome and melting point is lowest.Įthanedioic acid can form a maximum of 10 hydrogen bonds which is the most extensive, greatest amount of energy required to overcome hence it has the highest melting point. We can determine the maximum number of hydrogen bonds formed for each molecule by drawing out their lewis structures. Let's compare the melting points of ethanol, ethanoic acid and ethanedioic acid. Let's go through the factors affecting Hydrogen bond in this video, which are extensiveness and polarity.Ī molecule that can form more hydrogen bonds with more neighboring molecules will have more extensive hydrogen bonds, hence more energy is required to break more hydrogen bonds per molecule, and the melting/boiling point is higher.Įxtensiveness is related to the maximum number of hydrogen bonds a molecule can form with its neighbours. instantaneous dipole-induced dipole attraction (or dispersion forces or Van Der Waals forces) between non-polar molecules - permanent dipole-permanent dipole attraction between polar molecules - Hydrogen bond between polar molecules that have H-F, H-O or H-N bonds 
There are 3 types of intermolecular forces namely: Simple molecules have weak forces of attraction between them hence their melting and boiling points tend to be much lower as compared to other substances such as metals, ionic compounds and giant molecules.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
